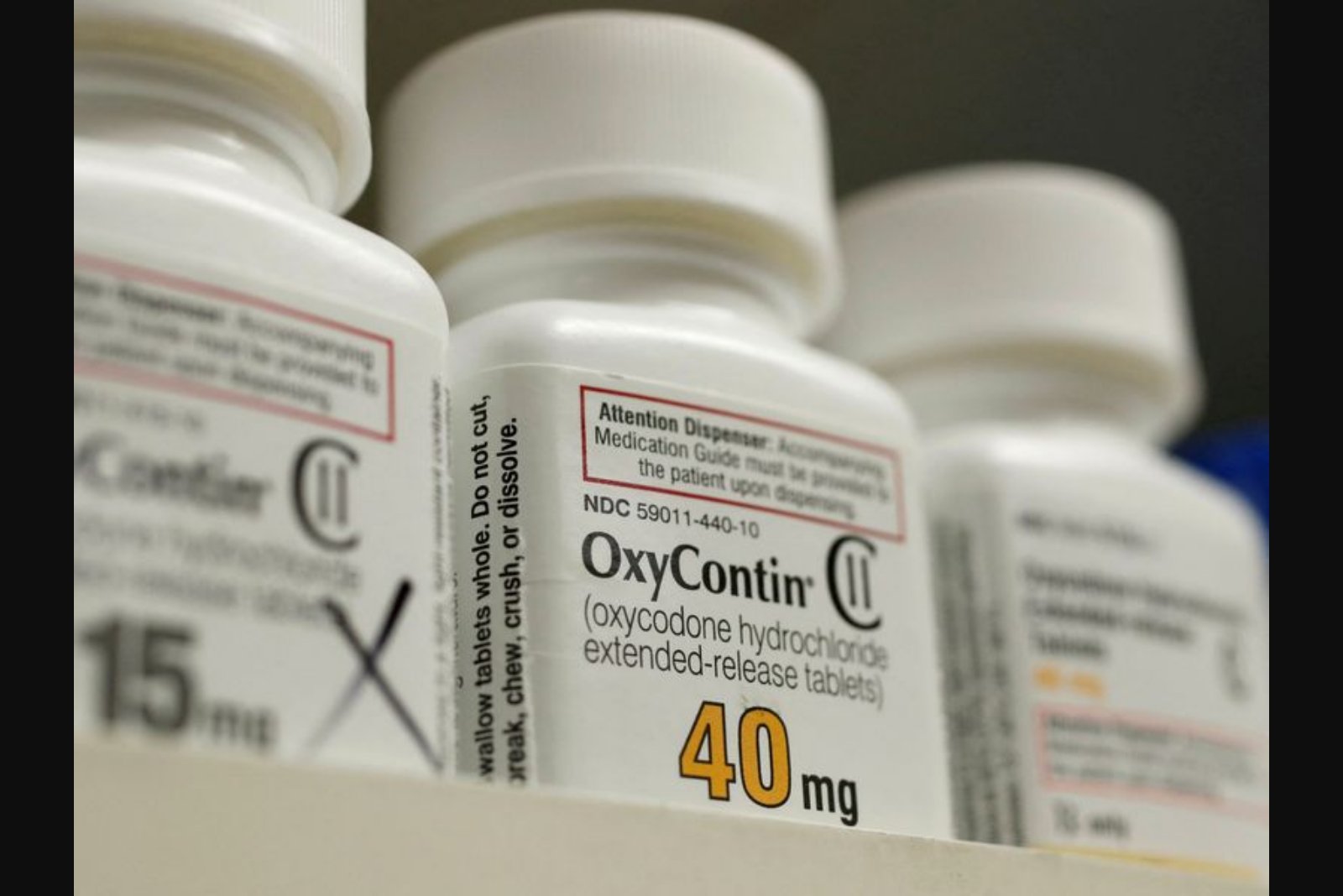
Purdue Pharma faces sentencing on Tuesday in federal court in Newark, New Jersey, after pleading guilty to charges that it deceived government regulators and paid kickbacks to doctors to expand sales of its opioid painkiller OxyContin. Prosecutors say the sentencing will conclude the criminal portion of a plea deal that enables the company to dissolve under Chapter 11 and allocate its remaining assets to a $7.4 billion settlement designed to provide relief to communities and individuals affected by the opioid epidemic.
Under terms disclosed in the plea agreement, Purdue agreed to $5.5 billion in criminal fines. Most of that sum will not be collected because of a 2020 arrangement with the U.S. Department of Justice that reduced the cash the Justice Department will seek to $225 million. That arrangement allows Purdue to direct the bulk of its remaining estate toward repaying creditors, who are primarily state and local governments that bore many of the public costs associated with the opioid crisis.
The sentencing had been scheduled for the prior week but U.S. District Judge Madeline Cox Arleo postponed it to give more members of the public a chance to speak about the company’s marketing practices for OxyContin and its contribution to the epidemic. Purdue issued a statement ahead of the hearing saying it recognized the importance of victims having the opportunity to be heard and that the company respected the court’s decision.
Victims and their advocates submitted letters to the court describing addiction, loss and long term suffering. Some of those who wrote urged the judge to reject the plea deal and to demand prison sentences for company executives and owners. Those appeals from victims underscore ongoing frustration with how the company’s prolonged bankruptcy has handled claims and compensation.
At the hearing in Newark, Judge Arleo is expected to accept the plea agreement and impose a $3.5 billion criminal fine plus $2 billion in criminal forfeiture, completing the $5.5 billion criminal obligation. Acceptance of that plea is one of the final legal steps required before the broader bankruptcy settlement can proceed.
Purdue’s Chapter 11 case has stretched for more than six years and included a long series of appeals that reached the U.S. Supreme Court. With sentencing completed, the company has said it remains on track to emerge from bankruptcy on May 1. Under the planned transition, Purdue would cease its prior commercial operations and be reconstituted as a new nonprofit entity focused on producing treatments for opioid addiction and medications to reverse overdoses.
As part of the plea agreement, Purdue admitted it paid kickbacks to physicians to drive OxyContin prescriptions and that it misled federal regulators about measures to prevent illegal use of the product. The latest criminal case did not include charges against any company executives or owners.
This is not Purdue’s first criminal admission. In 2007 the company pleaded guilty to misbranding and fraud charges tied to OxyContin marketing, acknowledging that it had marketed the drug as less addictive, less subject to abuse, and less likely to cause withdrawal symptoms than competing pain medications.
Context on implementation
The sentencing is framed as a necessary legal step to unlock the bankruptcy settlement process so that the company’s remaining assets can be channeled to creditors and to the settlement funds, including an $865 million pool earmarked for individuals affected by the crisis. Plaintiffs’ lawyers and Purdue have described the agreement as a milestone for victims, though some people harmed by opioids say the mechanics of the bankruptcy have left many claimants frustrated.
What happens next
- If the court accepts the plea, the company will receive the final court approvals needed to proceed with the planned corporate restructuring and asset distributions under the settlement framework.
- Following emergence from bankruptcy, Purdue intends to operate as a nonprofit manufacturer of addiction treatment and overdose reversal drugs rather than as a traditional opioid drugmaker.