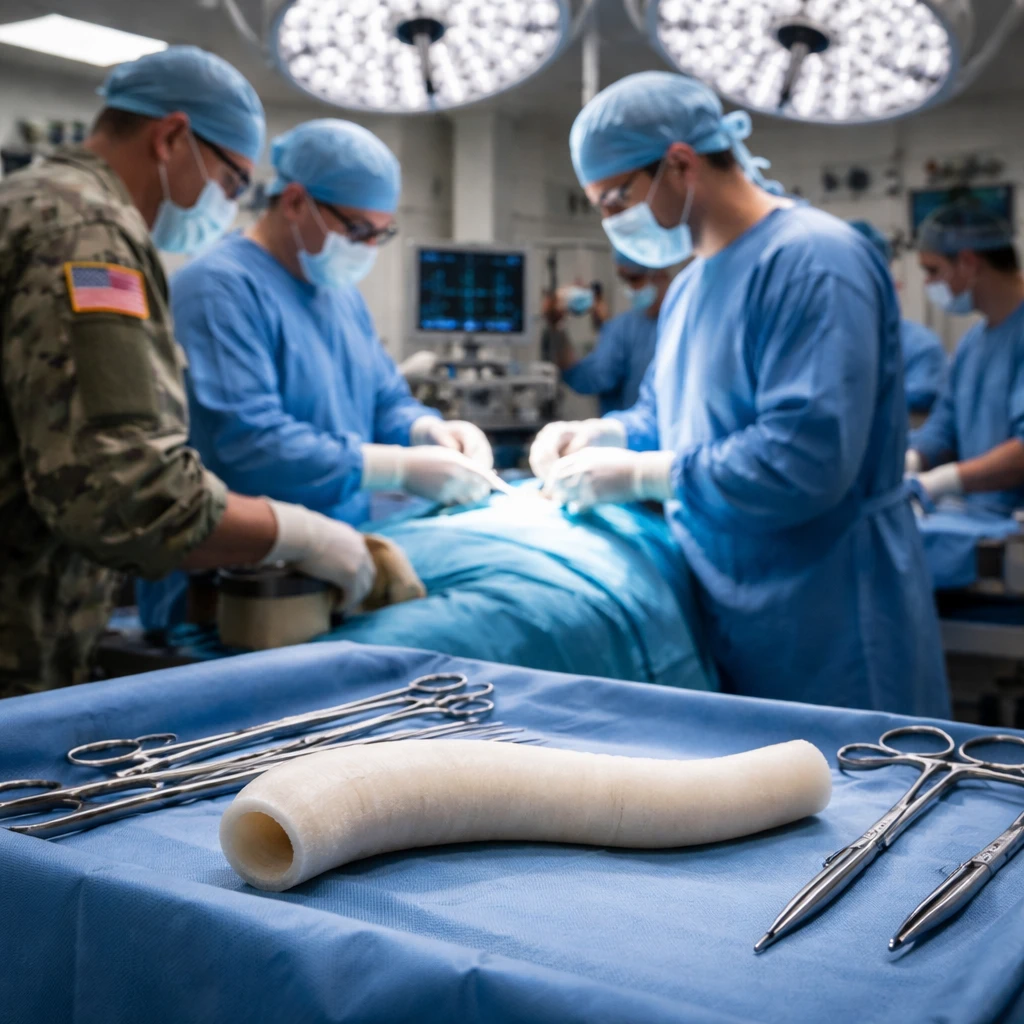
Shares of Humacyte Inc (NASDAQ: HUMA) jumped 11.4% on Monday after the company announced that the U.S. Department of Defense’s FY2026 Appropriations Act includes allocated funds to support bioengineered vascular repair technologies.
The federal appropriation marks a first-of-its-kind investment to evaluate and potentially incorporate biologic vascular repair options for military personnel suffering traumatic vascular injuries. That action follows language in the FY2026 National Defense Authorization Act that encouraged the Department of Defense to integrate FDA-approved breakthrough vascular repair technologies into care for injured service members.
Humacyte’s product Symvess is noted as the only FDA-approved human-derived bioengineered blood vessel currently available. The company says the graft has been deployed in a humanitarian program in Ukraine, where it treated patients with wartime injuries including gunshot wounds, shrapnel injuries, and blast trauma.
"America’s brave men and women in uniform deserve the very best care we can provide," said Laura Niklason, Founder and CEO of Humacyte. "This historic, first-of-its-kind federal investment will help ensure our soldiers continue to have access to cutting-edge treatments and state-of-the-art care whenever and wherever they need it."
Humacyte reported a retrospective observational study of sixteen patients treated with Symvess in Ukraine. According to the company, that review showed 100% survival, zero amputations, and zero conduit infections among the treated cohort, despite the severity of their wartime injuries.
Symvess is approved for adult use as a vascular conduit for extremity arterial injury when urgent revascularization is necessary to avoid limb loss and when an autologous vein graft is not feasible. The new DoD funding is intended to support evaluation and integration of such biologic vascular repair technologies into military trauma care pathways.
Investors reacted to the funding announcement with a notable bid for the stock, reflecting the market response to the federal appropriations language and the potential for a new buyer of approved vascular repair technologies within the defense medical system.
This report presents the company disclosures and regulatory status as stated by Humacyte and the appropriations and authorization language cited by the company. The information in this article is limited to those statements and the company-reported clinical observations.