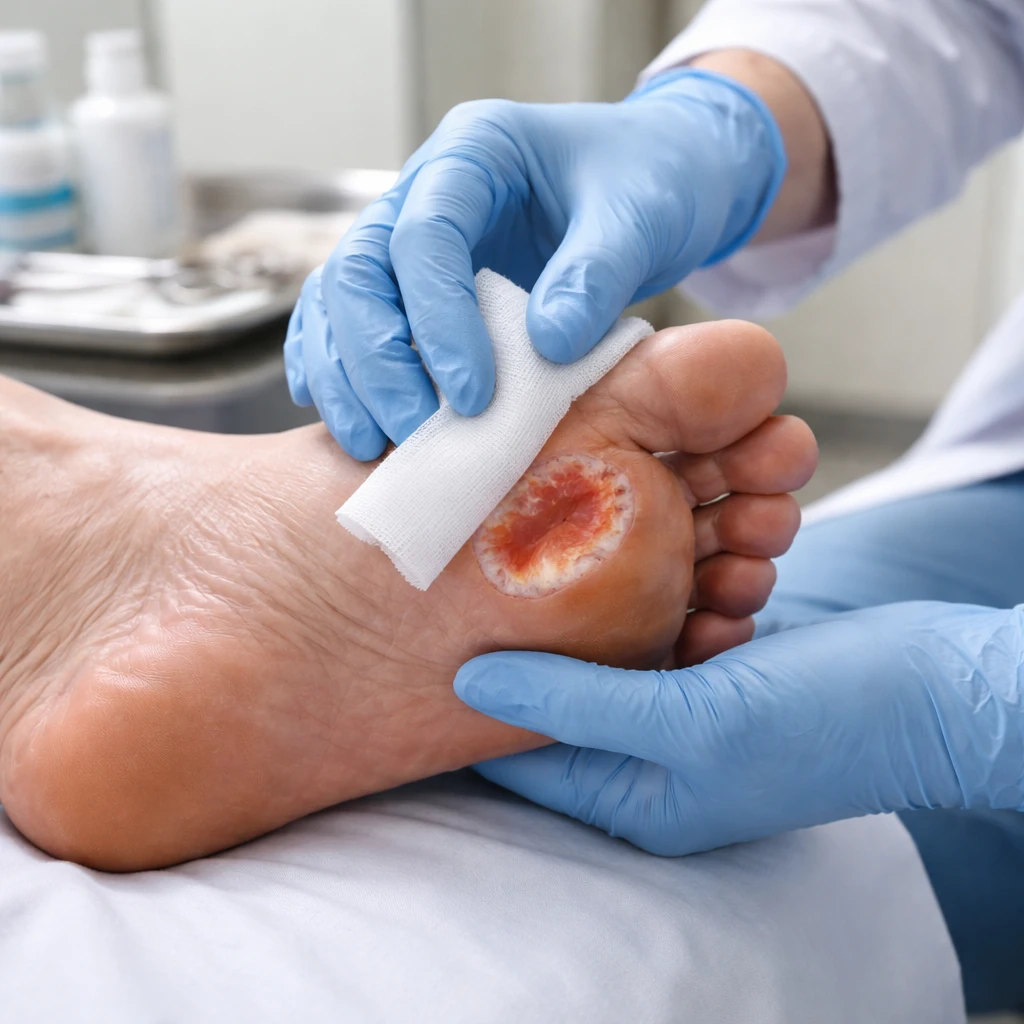
Organogenesis Holdings Inc. (NASDAQ:ORGO) experienced a 20% rise in its share price on Tuesday after the company disclosed that PuraPly AM met the primary endpoint in a randomized controlled trial assessing treatment for diabetic foot ulcers.
The prospective, multi-center study enrolled 170 patients and measured wound closure at 12 weeks. According to the company, the trial demonstrated a statistically significant improvement in wound closure when PuraPly AM was used in conjunction with standard of care compared with standard of care alone. The study was designed to evaluate the safety and efficacy of the treatment specifically in non-healing diabetic foot ulcers.
PuraPly AM is described by Organogenesis as a patent-protected product that combines a native, cross-linked collagen matrix with polyhexamethylene biguanide antimicrobial. The company states the formulation is intended to manage bioburden while supporting the wound-healing process.
Commenting on the outcome, Patrick Bilbo, Chief Operating Officer of Organogenesis, said the company was pleased to have achieved the primary endpoint in a study that underscores the clinical efficacy of PuraPly AM in managing non-healing diabetic foot ulcers. Bilbo noted that these wounds impose a significant burden on patients and are costly to the healthcare system. He added that publication of the results could strongly support PuraPly AM's inclusion in future coverage policies and reinforce its role within the healing paradigm.
Organogenesis indicated that the new trial data bolster existing clinical evidence that could support future coverage of the product. The company develops, manufactures and commercializes product solutions for the Advanced Wound Care and Surgical and Sports Medicine markets.
While the announcement focused on clinical outcomes and potential implications for coverage, Organogenesis did not provide additional details about next steps for publication timing or payer deliberations. The company likewise did not expand on commercial rollout plans tied to the new data in its statement.
Contextual note: The trial results as reported emphasize clinical performance at the 12-week mark and the company’s view that publication and the strengthened evidence base could influence coverage decisions. No further claims about regulatory status, reimbursement outcomes, or commercial returns were made in the company release.